

1 / 5










| Customization: | Available |
|---|---|
| Classification: | Physiological Functions of Diagnosis and Monitoring Equipment |
| Type: | Patient Monitor |
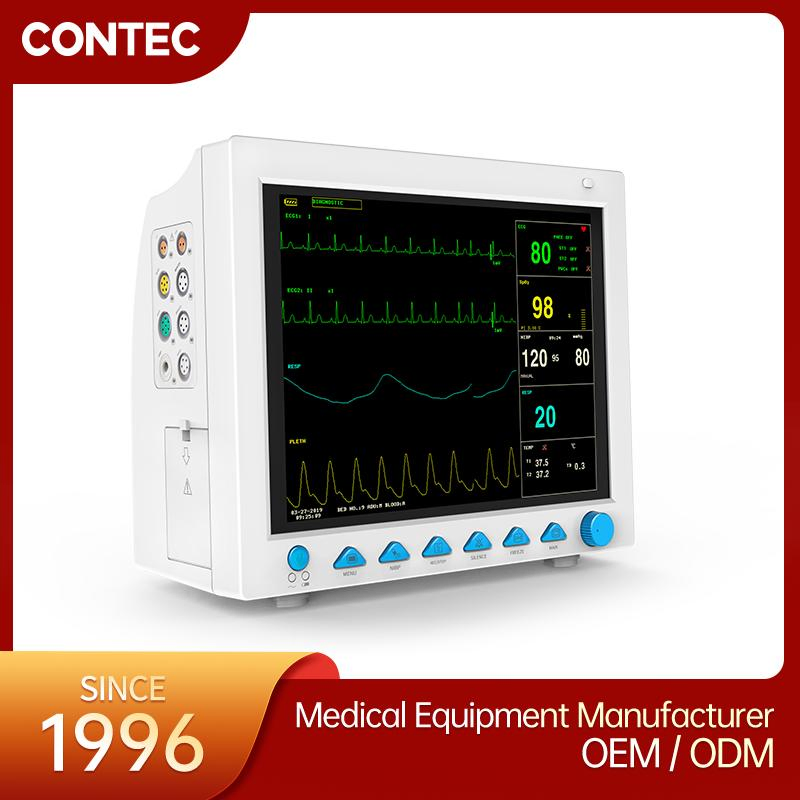
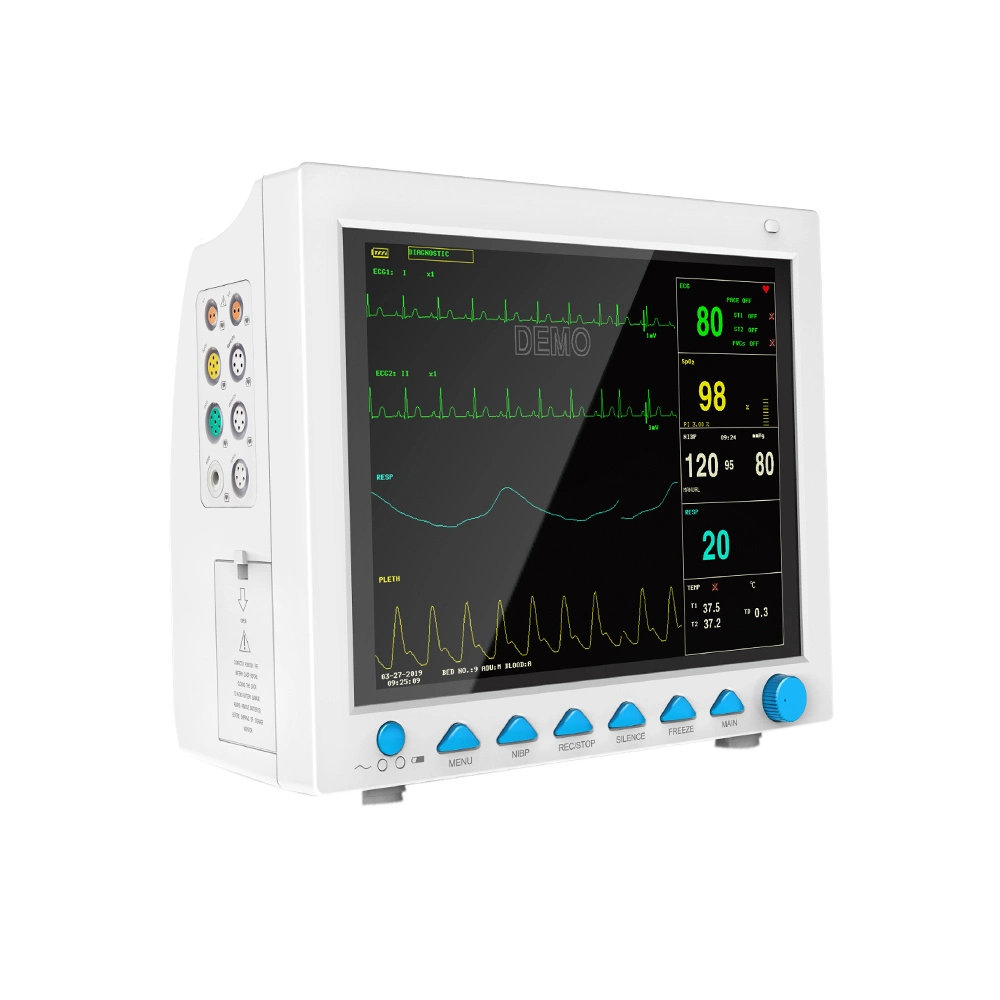
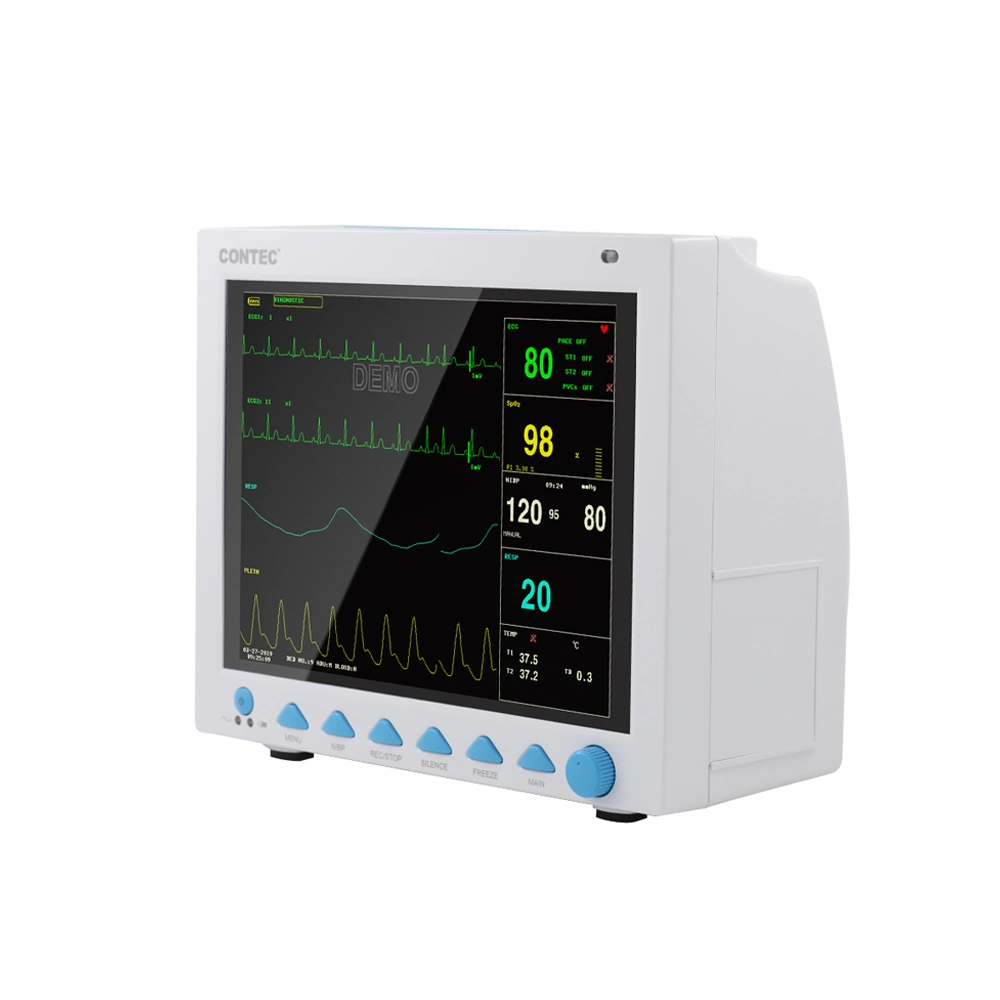
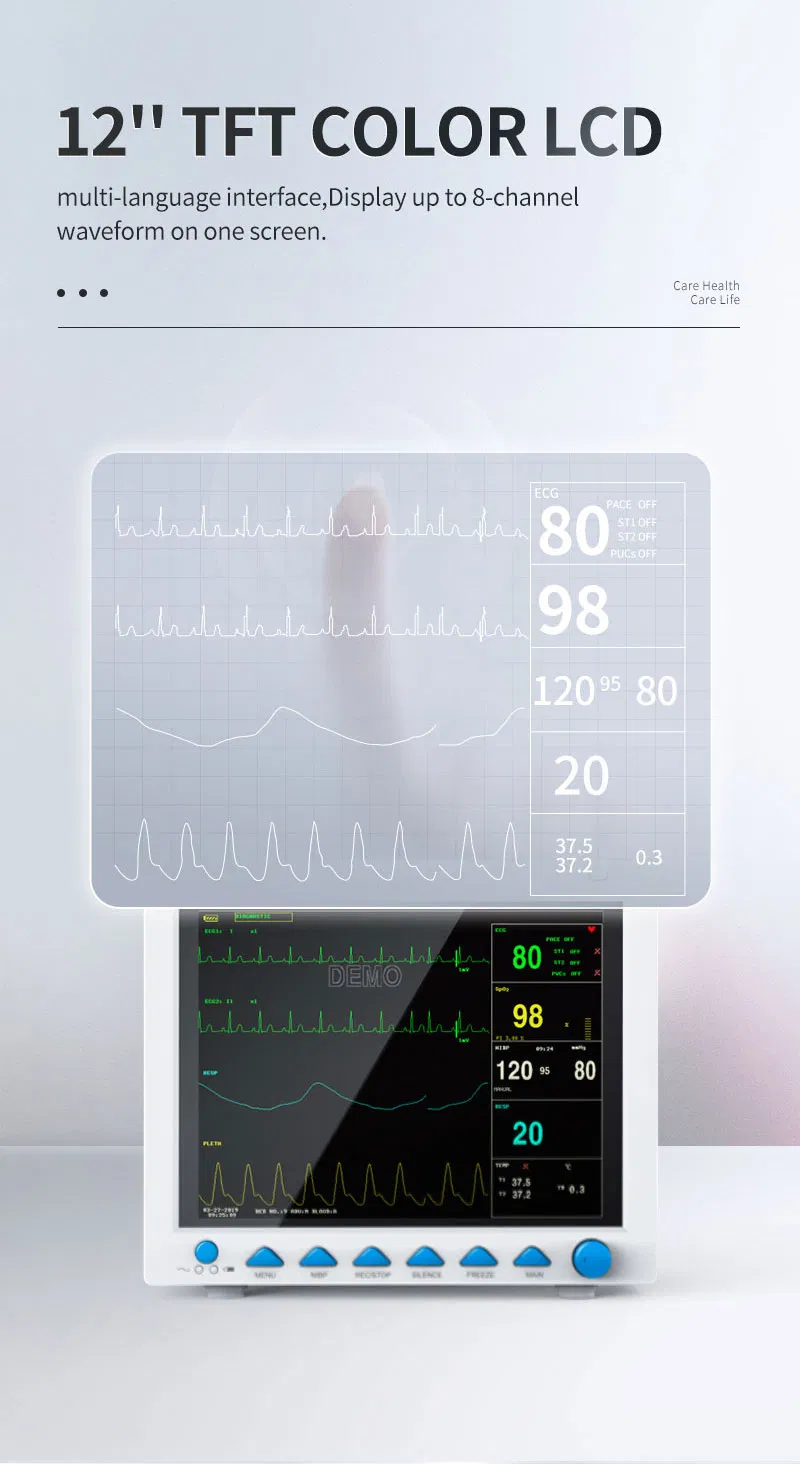
The CMS8000 Patient Monitor is a high-performance, portable multi-parameter device designed for all-round monitoring of adult, pediatric, and neonatal patients. With its high-resolution 12'' color TFT LCD, it provides clear visualization of vital signs in various medical environments.



| Parameter | Specification Details |
|---|---|
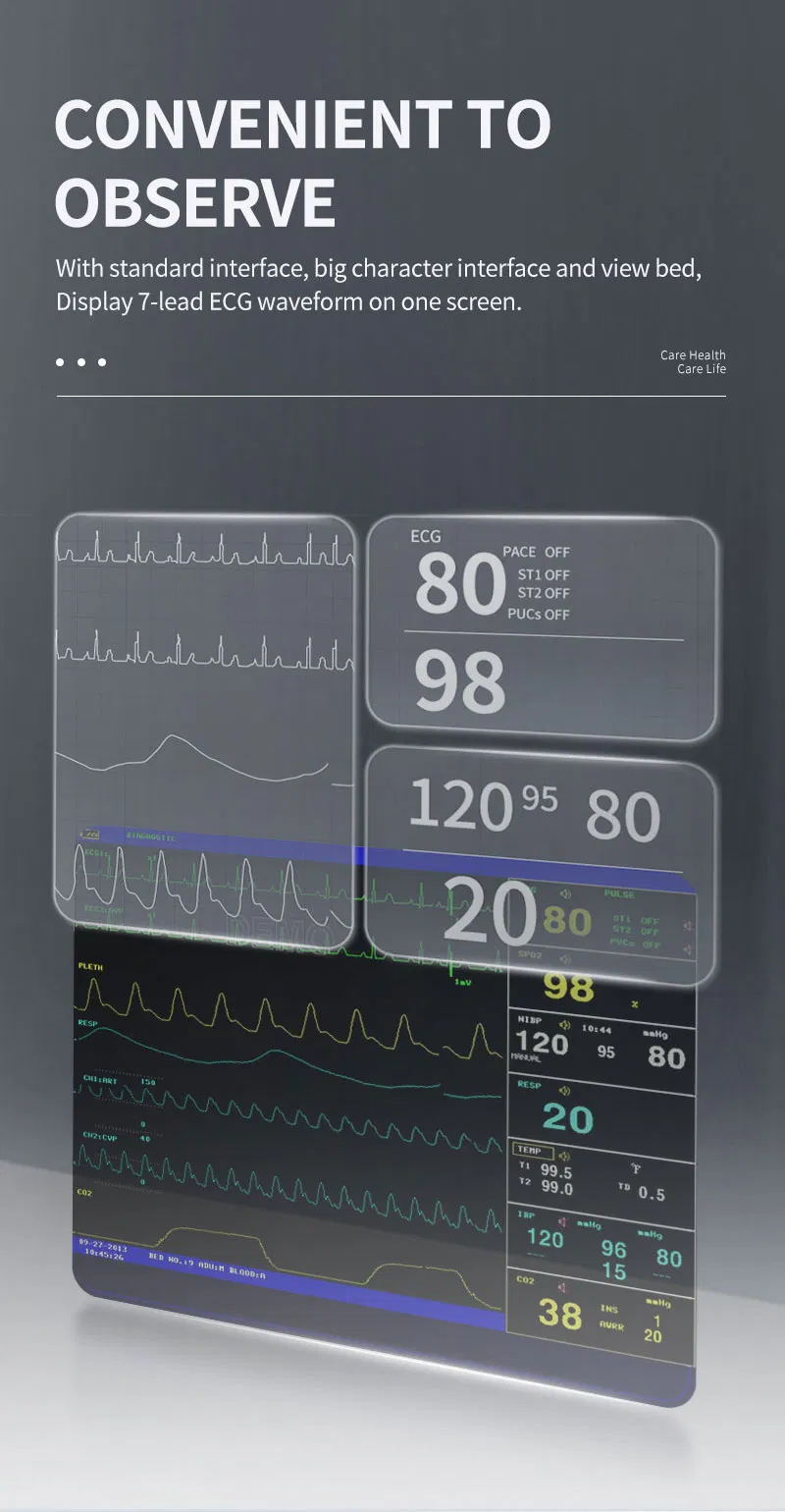
| ECG Lead Mode | 3-lead and 5-lead are optional; Lead Selection: I, II, III, avR, avL, avF, V |
| HR Range | 15 ~ 300 bpm |
| RESP Range | Adult: 0~120rpm; Pediatric/Neonatal: 0~150rpm |
| SpO2 Range | 0 ~ 100 % (Accuracy: 70% ~ 100% ±2%) |
| NIBP Method | Oscillometry; Manual, Auto, Continuous modes |
| TEMP Channel | Dual-channel; Range: 0 ~ 50°C |
| EtCO2 (Optional) | Sidestream or Mainstream; Range: 0~150mmHg |
| Power Requirement | AC 100~240V, 50/60Hz; Consumption: 150VA |
| Physical Specs | Dimension: 310×140×263(mm); Net Weight: 3.8Kg |








 Suffolk Medical
Suffolk Medical